When Ancel Keys discovered that high cholesterol concentrations in blood were associated with CHD in middle aged men, it highlighted the importance of this lipid that is actually in fairly low concentration within cells. It was known that cholesterol was a waxy substance but there was very little known about it except that it was the starting material for a group of steroid hormones, including estrogen and testosterone.
Little was known about the cholesterol in the blood until it was shown, by electrophoresis, to be carried by lipoproteins in blood. The lipoprotein families were finally isolated and characterized in the late 1940s using the newly developed ultracentrifuge (Gofman).
Gofman, J. W., Lindgren, F. T. & Elliot, H. (1949) Ultracentrifugal studies of lipoproteins of human serum. J. Biol. Chem. 179: 973–979.
http://www.jbc.org/content/179/2/973.full.pdf+html?sid=3464e673-7b4d-4ee7-b288-9d63bda6d824
The slide below shows that most cholesterol in blood is carried in Low Density Lipoprotein (LDL). High density lipoprotein (HDL) also carries cholesterol in blood, but much less than LDL does. We will discuss HDL later.
The following slide shows that Very Low Density Lipoprotein (VLDL) primarily delivers fatty acids to cells whereas LDL delivers cholesterol to cells. VLDL is secreted by the liver and exists in blood for several hours until most of the triacylglycerol molecules it carries are transferred to cells. About 50% of VLDL is converted to LDL, which mainly carries cholesteryl esters. LDL exists in blood for several days and it mainly delivers cholesterol to all cells except those in the brain, which LDL cannot enter due to the blood brain barrier.
Studies on the transport of fatty acids, cholesterol and lipoproteins using radioisotopes continued through the 1950s and 1960s (Fredrickson 1958), but there was still uncertainty about how lipoprotein particles interacted with cells.
Fredrickson, D. S. & Gordon, R. S. (1958) Transport of fatty acids. Physiol. Rev. 38: 585–630.
http://physrev.physiology.org/content/38/4/585.long
The mystery surrounding cholesterol and low density lipoprotein (LDL) persisted until two scientists unraveled the metabolism and function of LDL. Two new assistant professors, Joseph Goldstein and Michael Brown, at the University of Texas Southwestern Medical Center in Dallas, sought the metabolic reason for familial hypercholesterolemia (FH), a genetic disease where the concentration of cholesterol in blood is increased many fold over control, and where cholesterol deposits, called xanthomas, appear in the skin. These patients have heart attacks early in life, sometimes as early as six years old.
Drs. Goldstein and Brown cultured skin cells from patients who had the disease and compared cholesterol metabolism in them with its metabolism in fibroblasts from normal patients. They showed that adding LDL at very low concentrations to normal fibroblasts lowered the activity of a key enzyme in cholesterol synthesis, showing that LDL could affect cholesterol metabolism within cells.
FH fibroblasts did not react to treatment with LDL, so something was wrong with the ability of these cells to use LDL. Eventually, Drs. Goldstein and Brown determined that FH fibroblasts has a defective protein called the LDL receptor, which was involved in bringing LDL into cells. Cholesterol delivered to the cell with LDL is delivered to the cell using the LDL receptor and this cholesterol is capable of affecting (in this case, feedback regulating) cholesterol metabolism within the cell.
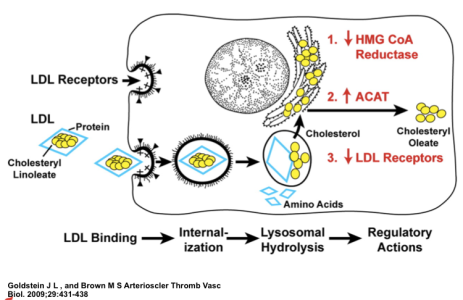
After purifying the LDL receptor, the laboratories of Drs. Goldstein and Brown went on to clone the gene for the LDL receptor, and show that the molecular defect in FH was a mutation in the LDL receptor protein. They later went on to show how the delivery of cholesterol to cells can modulate many pathways that control cholesterol metabolism in cells. This is shown on the fairly complex figure below.
The red writing within the cell diagram shows the major effects that occur in metabolism when LDL cholesterol is delivered to cells. First, the activity of HMG CoA reductase, an important regulatory enzyme in cholesterol synthesis, is decreased. Second, cholesterol esterification is increased in order to store extra cholesterol in the cell’s lipid droplets. Third, the numbers of LDL receptors are decreased in order to slow the influx of cholesterol into the cell through the LDL receptor pathway.
The discovery of LDL receptors provided insights into how lipoproteins are taken up by cells and gave rise to the eventual discovery of many other receptors and complex receptor mediated uptake mechanisms used by all cells. For this major discovery, Drs. Goldstein and Brown won the Nobel Prize in Physiology or Medicine on October 15, 1985 (See their picture below).
In addition to discovering the LDL receptor, Drs. Goldstein and Brown would go on to make many more amazing discoveries in cholesterol and lipid metabolism. These additional discoveries include deciphering: 1) the complex mechanism of sterol receptor element binding proteins (SREBPs), proteins that direct the transcription of genes involved in both cholesterol and fatty acid metabolism, 2) the cholesterol sensor in the membrane of the endoplasmic reticulum, and 3) the transport of cholesterol throughout the cell. Additionally, Drs. Goldstein and Brown have made many contributions to the understanding of many human disease states that involve lipid metabolism, including several hyperlipidemias and type 2 diabetes. Many researchers in the lipid field strongly believe that Drs. Goldstein and Brown deserve to be awarded a second Nobel prize in medicine.








Leave a short comment